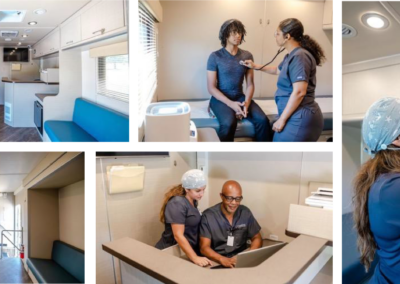
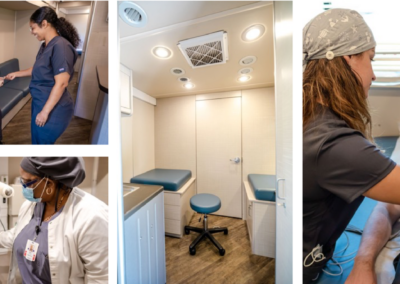
Community Research Sites
Local. Mobile. Global.
Mobile Solutions:
To improve clinical trials and accelerate the advancement of promising treatments, there is a need to provide improved patient access options and patient diversity. EmVenio’s mobile sites offer that possibility by providing more convenient access options for participants in the communities where they live and work. With sites more conveniently located to patients, travel, schedule, and daily life challenges are minimized resulting in improved willingness to participate and remain on studies. EmVenio works with our partners and communities to identify optimized locations to activate our state-of-the-art Mobile Community Research Sites, providing convenient access, increasing geographic reach and patient diversity:
- Provide convenient trial access solutions wherever trials are needed as a care option and across broad geographies
- Support a full range of protocol procedures, from standard to more complex
- Bring clinical trials into hard-to-reach communities and to underrepresented patient populations

Safety and Security Highlights
- Shore power and generator support: 24/7 backup supported by two generators
- 24/7 temperature monitoring: Automatic notification with temperatures are out of range
- Sharps containers and biohazard waste are managed and disposed of per EmVenio SOPs
- HEPA filtration throughout the mobile site and in each exam room
- Site location will provide sufficient physical security to allow it to stay on-site full-time
EmVenio’s Localized Approach
Reaches diverse participants
EmVenio actively engages underrepresented communities to ensure that a diverse range of populations is properly represented in clinical research.
44.7% of enrolled patients self-identified as minority ethnicity in completed studies
Expands the reach of clinical trials
EmVenio has an in-house team of clinicians and Principal Investigators that allow us to reach a broad geographical area and recruit diverse communities.
Improves the Trial Participant Experience
EmVenio has a patient-centric approach, meeting participants where they live and work to improve participant compliance, retention, and experience.
>92% Retention Rate
Helps Partners Meet Trial Goals
- EmVenio offers customizable solutions based on protocol design, patient needs, and community demographics. We collaborate with partners to determine the best mix for each study’s unique requirements.
- Providing certainty around enrolment and diversity, EmVenio helps accelerate study completion
Provides Expert Clinical Teams
- EmVenio clinicians have a full spectrum of licensure levels, allowing us to expand access, accelerate timing of study completion, deliver high-quality data metrics, and improve trial participant experiences. Clinical teams can include, NP, RN, LPN, Phlebotomist, PA, MD, etc.
- EmVenio PIs have overseen research on over 900 studies.
Ensures Quality and Compliance
EmVenio adheres to all GCP and ICH guidelines, with clear Quality policies and procedures in place to support participant safety.
Provides Robust Personnel Training
Our robust training curriculum is designed to help research professionals at all levels to better serve participants and clients, focusing on key competencies including: Scientific Concepts and Research Design, Data Management and Informatics, Ethical and Participant Safety Considerations and much more.
Fully digitally-enabled
- eConsent
- eSource
- eISF
“Greetings New Creation. I just wanted to give a spiritual shout out to the Research Company, EmVenio who’s using our parking lot. Whenever something is happing either at the building or in our community, they reach out to me and say, what can we do to help? This morning, Cal texted me and said, because of the tornados, whatever the church is doing to assist LR’s surrounding area, we want to partner with you. That is so awesome. Please pray for Cal and his team.”
Pastor D.
New Creation Bible Church
Little Rock, Arkansas